Hi, Hope you are well. In this article, we will study what is Electrochemical Deburring? and its Definition, Parts or Construction, Working principles, Application, Advantages, and Disadvantages in detail.
Note: PDF you can download at the end of the articles.
The use of electrochemical machining started in the early 1900s. Experiments were made till 1929. In 1956 electrochemical machining became commercial and the use of electrochemical energy to remove metal became common.
The exact history of the development of electrochemical deburring is known, yet we can say that with growing demands of accurate products there was a need to discard burrs after machining.
Electrochemical deburring was thus developed as a branch of electrochemical machining to eliminate burrs to obtain better-finished products.
What is electrochemical chemical deburring?
The word deburring means removal of burr from the source of any workpiece to provide a smooth finished surface. The term electrochemical means the mode of energy used for deburring. Together electrochemical chemical deburring refers to a machining process in which burrs are removed using electrochemical energy.
A special type of tool is used for this operation which is called a deburring tool. Like any other electrochemical process, in electrochemical deburring also there is no contact between the deburring tool and the workpiece.
It acts as a reverse electroplating process. In electrochemical deburring, the tool acts as the cathode and the workpiece acts as the anode.
Both the tool and the workpiece are kept in a flowing electrolyte medium. ECD is also known as electrolytic deburring due to the use of electrolytes. It is a fast and easy process.
Note: A burr is a small unwanted advancement of metal on the edge of the workpiece which is mechanically machined.
Need of Electrochemical Deburring:
Removal of burrs is considered a serious problem in Many industries which deal with high accuracy.
- A burr can sometimes have sharp edges which may harm the operator or a worker hence it is important to remove it.
- It can cause cracks on the surface of the mating parts. As the pressure increases when the area of contact decreases.
- It also lessens the beauty of the workpiece.
- Electrochemical deburring is also important for deburring hard metals.
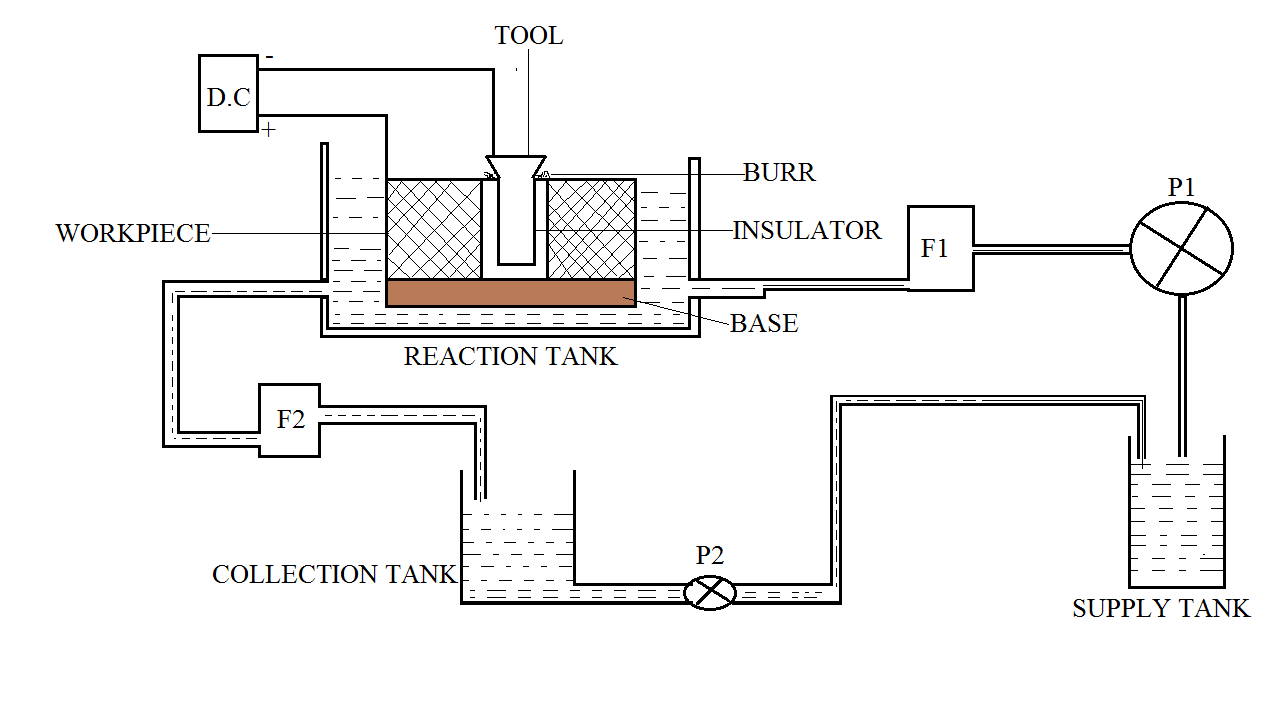
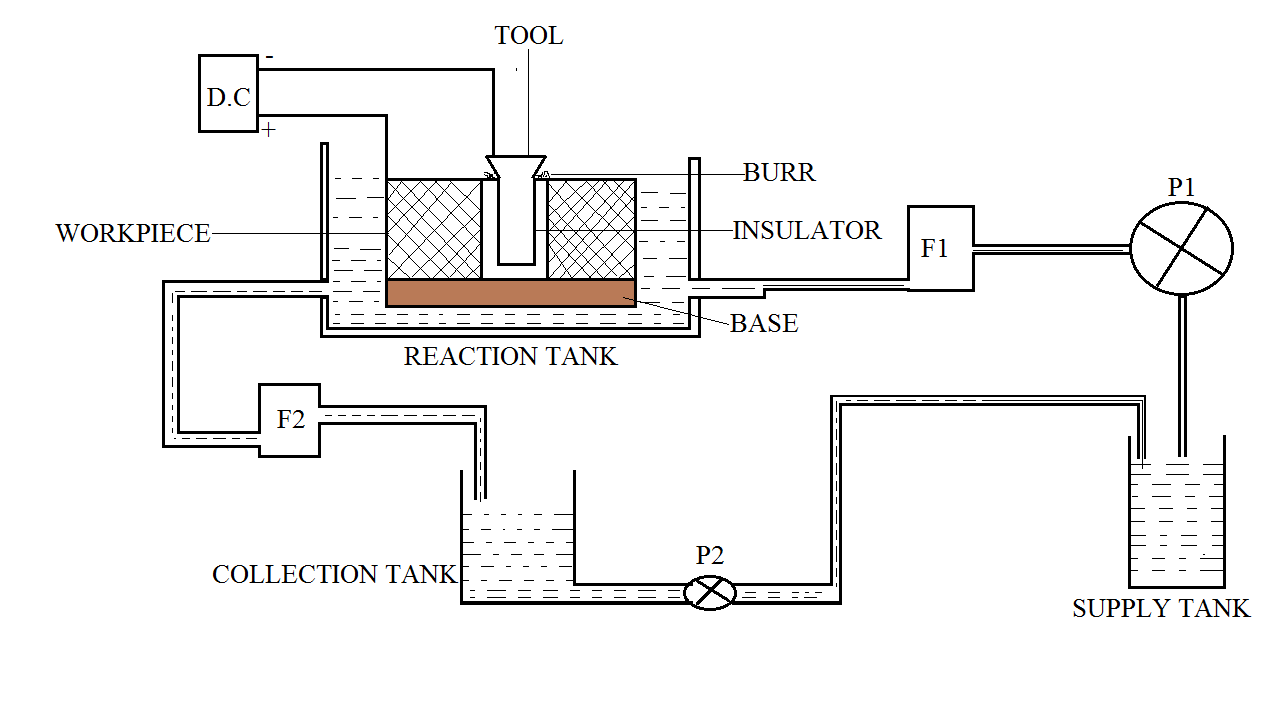
Components of Electrochemical Deburring:
Different Components or Parts of an electrochemical deburring setup are as follows:
- Supply tank
- Pumps
- Collection tank
- Reaction tank
- DC power supply
- Base
- Electrolyte and
- Tool
Supply tank:
The tank carrying electrolyte for its supply is called a supply tank.
Pumps:
There are two pumps P1 and P2. P1 is used to supply electrolytes to the reaction tank and P2 is used to supply the electrolyte from the collection tank to the supply tank.
Collection tank:
The tank which collects the filtered electrolyte is called a collection tank.
Reaction tank:
The area or the container carrying the tool-workpiece and the electrolyte are called a reaction tank. The electrochemical reaction between the workpiece and the tool takes place in the reaction tank.
The tank is designed in such a way that there is a constant flow of electrolytes in the tank to carry the slag.
DC power supply:
The voltage values of the DC power supply used in electrochemical deburring are low. But the current value is high, this promotes faster removal of metal from the surface of the workpiece.
Base:
The base here is made of a conducting material that is used to keep the workpiece stable. The base also connects the two workpieces electrically. DC supply is given to the base which connects the two workpieces.
Electrolyte:
The electrolyte is a solution of simple salt with water. It is generally a conductive solution of Sodium chloride and Sodium nitrate in water.
For general applications Sodium, nitrate and water are mixed in the ratio of 2:1. The temperature of the electrolyte is maintained at 20 degree Celsius for best results. The mixture of several salts is used in the case of metals like titanium.
Tool:
The Tool is the most important component of an electrochemical deburring setup. For various purposes, various different tools are used.
It consists of a conductometric which is insulated from the outside. The tool acts as the cathode which is connected to the negative terminal of the DC power supply.
A gap of 0.5-1 mm should be maintained between the workpiece and the tool. The basic procedure to design an electrochemical deburring tool is as follows.
- If the height of the workpiece is 15mm and the height of the burr is 2mm.
- Then the tool has to be designed in such a way that its height should be more than 15 + 2 = 17mm.
- It should be insulated till 15mm so that the material removal takes place above 15mm which removes the burr.
Working of the Electrochemical Deburring:
Before proceeding with the working one must understand the working principle of electrochemical deburring.
The Electrochemical deburring works on the principle of reverse electroplating. According to Faraday’s law of electrolysis, the amount of metal displaced is directly proportional to the electric current. When a high current is applied to the electrochemical deburring setup the material removal takes from the workpiece to the tool. The removal takes place from the gap between the tool and the workpiece. The material does not get deposited on the tool, it flows away due to the flow of the electrolyte. In this way, a highly finished surface is obtained.
Working step by step:
The workpiece is kept on the base and the tool is positioned between the workpieces.
The workpiece is connected to the positive terminal of the DC power supply. And the tool is connected to the negative terminal of the DC power supply. The pump is activated and the flow of electrolytes is started.
The electrolyte reaches the reaction tank by passing through a filter. Then the DC power supply is switched on and the reaction starts.
Electron transfer takes place between the workpieces and the electrolyte. The electron transfer results in the removal of burrs from the surface of the workpiece.
The remaining electrolyte is made to flow through the filter (F2) into the collection tank. From the collection tank, the electrolyte is again supplied to the supply tank. And the process is repeated.
Once the burr is removed the power supply is switched off and the workpiece is removed.
Working Video of the Electrochemical Deburring:
Advantages of Electrochemical Deburring:
The following merits or advantages of Electrochemical Deburring are:
- Electrochemical deburring is a highly accurate process which gives excellent surface finish.
- Heat generation is negligible.
- There are no thermal stresses developed in the workpiece.
- No wear of tool takes place.
- The efficiency is more.
- Faster process increases productivity of the plant.
- Quality products are manufactured.
Disadvantages of Electrochemical Deburring:
The following demerits or disadvantages are:
- High initial cost of the equipment.
- Different tool must be designed for different workpieces.
- Complex process.
- Highly skilled operator is required.
- Only the workpieces that conducts electricity can be machined.
Applications of Electrochemical Deburring:
The following application is:
- It is used for deburring of gears.
- It is also used for removing sharp edges from highly precise equipment.
- Also be used for surface finishing of hard materials.
Related Resources:
- Electrochemical Grinding
- Abrasive Jet Machining
- Ultrasonic Machining
- Laser Beam Machining
- Electrochemical Machining
- Grinding Machine
- Lathe Machine
- Milling Machine
Reference:
- Sciencedirect.com research paper on Deburring
- Youtube Video: SME Media
Conclusion:
So here we have studied Electrochemical Deburring in detail. Let me know what else I can help you in this topic or any other topic?. You can check our another article I am sure that can boost your knowledge. Till then Thank you for reading.







Discussion about this post